 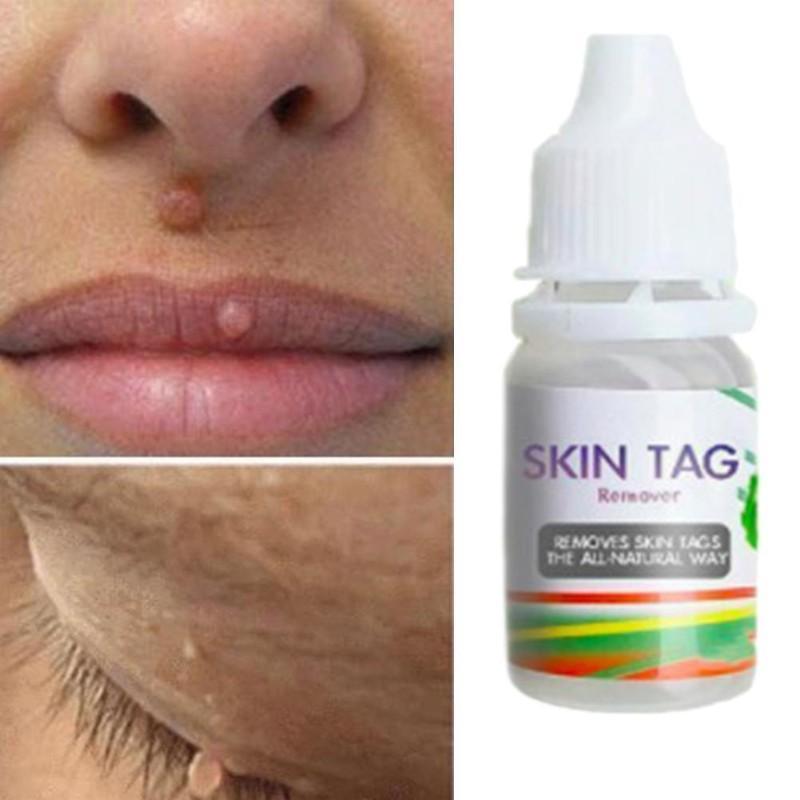
We made several observations about product packages and descriptions. However, some injuries were associated with products listing multiple botanical ingredients (including homeopathics) or no ingredients at all. Suspect products frequently contained concentrated SA, and reported injuries are consistent with SA’s capacity to destroy cutaneous epithelium. Many reviews were posted in 2021, indicating recent and ongoing product use. Amazon consumer reviews are a novel data source that allowed detection of skin injuries associated with unapproved mole and skin tag removers. Traditional data sources used by the FDA to identify drug safety issues include spontaneous reporting databases like FAERS and case reports in the medical literature. Figure 1 depicts representative injuries. A dermatologist advised one person to have a skin graft. Several required antibiotics (4), wound care (3), and/or hospital care (3). Twelve cases stated or implied that they sought medical care. When reported, time to symptom onset ranged from immediate (5) to within 24 hours (4), to 2 to 3 days (2) to one week (1). Seven consumers developed skin injury following a single application, which in several instances was described as a small amount, including two who applied the product with a toothpick. Fourteen injuries involved the face, including four adjacent to the eye. Adverse events (not mutually exclusive) included burn (30), scar (15), pain/discomfort (13), ulcer (13), infection/cellulitis (4), dermatitis/rash (2), and swelling (2). Reasons for use included moles (12), skin tags (12), anal skin tag (1), and unspecified lesions (15). canadensis (6), multiple botanical or “homeopathic” ingredients (5), calcium oxide (2), and unspecified ingredients (7). We identified 38 cases summarized in Table 1, including 30 (sex/age uncertain) from and 8 (4 females, 3 males, 1 not reported median age 51 years) reported to the FDA. We searched for reviews on other large online retailer websites, but none had the quantity and depth of information that was available on. We hypothesized that consumer reviews might augment adverse event reports submitted to FDA. 13, 14 We found no articles utilizing consumer reviews to detect adverse events associated with skin products. For example, there are publications that used online consumer reviews to identify adverse events associated with dietary supplements and to determine consumer perceptions about skin products. 
Online consumer reviews are a potential data source for pharmacovigilance. 12 The OTC Monograph for wart remover drug products requires inclusion of the following warning in the Drug Facts Label: “Do not use on moles, birthmarks, warts with hair growing from them, genital warts, or warts on the face or mucous membranes.” 12 
11 The FDA allows inclusion of SA in topical OTC drug products marketed pursuant to OTC Drug Monographs at concentrations ranging from 0.5%–40%, depending on the indications, which include acne (0.5%–2%), psoriasis (1.8%–3%), and hyperkeratotic conditions like corns, calluses, and warts (5%–40%). 
SA is a keratolytic at concentrations between 3%–6%, becoming more destructive at higher concentrations. These cases prompted our further review of this issue. They applied the product to lesions on the face, neck, torso, and buttocks as directed and sustained burns with resulting scars. Ingredients included concentrated SA 25%. The FDA has received cases from consumers who reported purchasing mole and skin tag remover products from. canadensis, including cosmetic disfigurement, and death due to progression of inadequately treated malignancy. 7, 8 There are also case reports describing harm from S. 6 Two additional published case reports described scarring following the use of an unapproved mole and skin tag remover containing cashew and fig, which may cause skin irritation, and greater celandine, a plant containing the escharotic alkaloid sanguinarine, also found in Sanguinaria canadensis. 2 – 5 Awareness of these products will allow dermatologists to counsel patients to avoid them and seek appropriate interventions instead.Ī recent publication described skin injury related to a salicylic acid (SA)-containing mole and skin tag remover product. The FDA has warned against using over the counter (OTC) products marketed for self-removal of moles or skin tags and has also issued warning letters to firms who sold such products. 1 There are no FDA-approved drugs indicated to treat moles or skin tags. The American Academy of Dermatology (AAD) advises the public to “never try to remove a mole at home” due to risks of scarring, infection, and the possibility of malignancy in the mole of concern. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
